What is NeuroStar Advanced Therapy (TMS)?
During a NeuroStar treatment session, a magnet similar in strength to that used in a magnetic resonance imaging (MRI) machine is used to stimulate nerve cells in the area of the brain thought to control mood. These magnetic pulses may have a positive effect on the brain's neurotransmitter levels, making long-term remission possible.
Take the first step
to overcoming your depression today.
Schedule a free consultation with our NeuroStar Coordinator to see if this treatment is right for you.
Treatment with NeuroStar Advanced Therapy is easy
Therapy sessions are conducted in your NeuroStar doctor's office
You can return to normal activities right away
Your are awake during the treatment
There are no negative effects on memory or sleep
It's covered by most health insurance plans, including Medicare
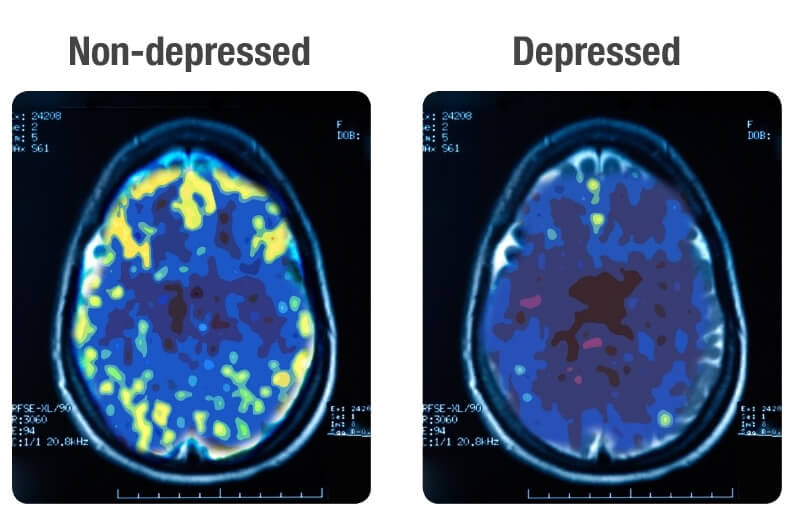
A PET Scan measures vital functions such as blood flow, oxygen use, and blood sugar (glucose) metabolism.
Source: Mark George, M.D. Biological Psychiatry Branch Division of Intramural Research Programs, NMH 1993
How NeuroStar® TMS Therapy works
Here's what you can expect from a NeuroStar Advanced Therapy (TMS) session:
NeuroStar ® Therapy Insurance Coverage
Since the FDA clearance of TMS in 2008, insurance coverage for eligible patients has increased significantly. Currently, there are over 60 coverage policies for TMS, including most Medicare contractors. Although TMS is not a first line of treatment, it is an alternative option for those who are not responding to or cannot tolerate medications. Call us today to speak directly with a NeuroStar Coordinator about your specific insurance plan, coverage, and eligibility. Click Here to contact us to see if your insurance company offers coverage for TMS treatments.
Professional Athlete Drew Robinson’s Journey with NeuroStar
Frequently Asked Questions
What is Transcranial Magnetic Stimulation?
How does TMS work?
How long is TMS treatment?
Does it Hurt?
Is TMS Therapy covered by my insurance?
Is TMS Therapy a good alternative for patients who cannot tolerate the side effects of antidepressant medications?
Is TMS Therapy like other alternative therapies that use magnets to treat some illnessess?
NeuroStar ® Patient Stories
Clinical Trials and Academic Studies
Carpenter LL, et al. (2012). Transcranial Magnetic Stimulation (TMS) for Major Depression: A Multisite, Naturalistic, Observational Study of Acute Treatment Outcomes in Clinical Practice. Depression and Anxiety, 29(7):587-596. www.ncbi.nlm.nih.gov/pubmed/22689344
George MS, et al. (2010). Daily Left Prefrontal Transcranial Magnetic Stimulation Therapy for Major Depressive Disorder: A Sham-Controlled Randomized Trial. Arch Gen Psychiatry, 67(5):507-516. www.ncbi.nlm.nih.gov/pubmed/20439832
Dunner DL, et al. (2014). A Multisite, Naturalistic, Observational Study of Transcranial Magnetic Stimulation (TMS) for Patients with Pharmacoresistant Major Depressive Disorder: Durability of Benefit Over a 1-Year Follow-Up Period. J Clin Psychiatry. 75(12):1394-1401. www.ncbi.nlm.nih.gov/pubmed/25271871
O’Reardon JP, et al. (2007). Efficacy and Safety of Transcranial Magnetic Stimulation in the Acute Treatment of Major Depression: A Multisite Randomized Controlled Trial. Biol Psychiatry, 62(11):1208-1216. www.ncbi.nlm.nih.gov/pubmed/17573044
The NeuroStar Advanced Therapy System is indicated for the treatment of depressive episodes and for decreasing anxiety symptoms for those who may exhibit comorbid anxiety symptoms
in adult patients suffering from Major Depressive Disorder (MDD) and who failed to achieve satisfactory improvement from previous antidepressant medication treatment in the current episode.
The NeuroStar Advanced Therapy system is intended to be used as an adjunct for the treatment of adult patients suffering from Obsessive-Compulsive Disorder (OCD).
NeuroStar Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar Advanced Therapy is right for you. Patients’ results may vary.
Visit neurostar.com for full safety and prescribing information.


